Spine Cage Manufacturing Insights: Innovations and Advanced Techniques Explained
Spine cage manufacturing is a specialized area within medical device production focused on creating small implantable structures used in spinal surgery.
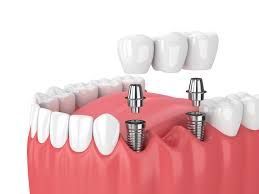
These implants, often referred to as spinal implant cage or spine cage implants, are designed to support the spine after damaged discs are removed. They help maintain proper spacing between vertebrae and promote bone fusion over time.
The concept originates from spinal fusion procedures, where surgeons aim to stabilize the spine and reduce pain caused by conditions such as degenerative disc disease. Over the years, spinal cage manufacturers have developed a range of designs and materials, including titanium spine cage and PEEK spinal cage options, to improve patient outcomes.
Modern interbody fusion cage manufacturing combines engineering precision, biomaterial science, and advanced machining techniques. These implants are used in procedures such as lumbar interbody fusion cage placement or cervical spine cage implants, depending on the affected region of the spine.
Importance
Spinal health plays a critical role in mobility and overall quality of life. When spinal discs deteriorate or become damaged, they can cause chronic pain, nerve compression, and reduced movement. Spine cage implants for degenerative disc disease are designed to address these issues by stabilizing the affected area.
The importance of spinal fusion cage for surgery lies in its ability to:
- Restore spinal alignment and maintain disc height
- Support bone growth between vertebrae
- Reduce pressure on surrounding nerves
- Improve structural stability after disc removal
For patients undergoing spinal procedures, the design and quality of spinal surgery implant devices can influence recovery and long-term outcomes. Minimally invasive spine cage systems are also becoming more common, allowing surgeons to perform procedures with smaller incisions and potentially faster recovery times.
Custom spine cage manufacturing has further expanded the field by enabling implants tailored to individual patient anatomy. This approach is particularly useful in complex cases where standard implants may not provide an ideal fit.
Recent Updates
Recent developments in spine cage manufacturing insights show a shift toward precision, personalization, and improved material performance. Between 2024 and 2026, several trends have shaped the field.
Advanced Materials
Manufacturers are increasingly using hybrid materials that combine the strength of titanium with the flexibility of polymers like PEEK. These combinations aim to mimic the natural properties of bone more closely.
- Titanium spine cage designs now often feature porous structures to encourage bone integration
- PEEK spinal cage models are being enhanced with surface coatings to improve fusion rates
Additive Manufacturing
3D printing has become an important technique in interbody fusion cage manufacturing. It allows for:
- Complex geometries that are difficult to achieve with traditional machining
- Custom spine cage manufacturing tailored to patient-specific anatomy
- Reduced material waste during production
Minimally Invasive Design
Minimally invasive spine cage systems are evolving to support smaller surgical approaches. These designs focus on:
- Compact आकार for easier insertion
- Enhanced stability despite reduced size
- Compatibility with advanced surgical tools
Smart Implant Research
There is ongoing research into implants that can monitor healing progress. While still developing, these innovations may eventually allow real-time tracking of spinal fusion.
Laws or Policies
Spinal implant manufacturing is governed by strict regulations to ensure patient safety and product reliability. These rules vary by country but generally follow similar principles.
Regulatory Frameworks
In many regions, spinal fusion cage manufacturers must comply with medical device regulations such as:
- Classification systems based on risk level
- Pre-market approval or clearance processes
- Clinical evaluation and testing requirements
For example, in India, spinal surgery implant devices fall under the oversight of the Central Drugs Standard Control Organization (CDSCO). Manufacturers must ensure that their products meet quality and safety standards before they can be used in clinical settings.
Quality Standards
Manufacturers typically follow internationally recognized standards, including:
- ISO guidelines for medical device quality management
- Biocompatibility testing standards
- Sterilization and packaging requirements
Traceability and Documentation
Each spinal implant cage must be traceable throughout its lifecycle. This includes:
- Batch identification
- Manufacturing records
- Post-market monitoring
These policies help ensure that any issues can be identified and addressed efficiently.
Tools and Resources
Spine cage manufacturing involves a range of tools and platforms that support design, testing, and production.
Design Software
Engineers use advanced software to create and refine implant designs:
- CAD tools for modeling spinal cage implants
- Simulation software to test stress and load distribution
- Finite element analysis for predicting performance
Manufacturing Equipment
Modern facilities rely on specialized equipment for precision production:
- CNC machines for shaping titanium spine cage structures
- 3D printers for additive manufacturing
- Surface treatment systems for improving implant integration
Testing and Validation Tools
Before use, implants undergo rigorous testing:
- Mechanical testing उपकरण for strength and durability
- Imaging tools to evaluate structural integrity
- Biocompatibility testing platforms
Reference Platforms
Educational and technical resources help professionals stay informed:
- Medical journals covering spinal fusion research
- Regulatory websites outlining compliance requirements
- Industry publications discussing innovations in spinal implant cage technology
Key Materials Used in Spine Cage Manufacturing
The choice of material plays a crucial role in implant performance. Below is a simplified comparison of commonly used materials:
| Material Type | Key Features | Common Use Cases |
|---|---|---|
| Titanium | High strength, durable, biocompatible | Load-bearing implants, porous designs |
| PEEK | Lightweight, flexible, radiolucent | Imaging-friendly implants |
| Hybrid (Ti + PEEK) | Combines strength and flexibility | Advanced interbody fusion cage designs |
| Ceramic Coatings | Enhances bone integration | Surface-treated implants |
Each material is selected based on factors such as patient condition, implant location, and surgical requirements.
FAQs
What is interbody fusion cage manufacturing?
Interbody fusion cage manufacturing refers to the process of designing and producing implants used to stabilize the spine during fusion procedures. These cages are placed between vertebrae to support bone growth and maintain alignment.
How do spinal cage implants help in surgery?
Spinal cage implants provide structural support after a damaged disc is removed. They help maintain proper spacing between vertebrae and promote bone fusion, which stabilizes the spine over time.
What is the difference between titanium spine cage and PEEK spinal cage?
A titanium spine cage is मजबूत and often used for load-bearing applications, while a PEEK spinal cage is lighter and allows better visibility in imaging scans. The choice depends on surgical needs and patient factors.
Are minimally invasive spine cage systems widely used?
Minimally invasive spine cage systems are becoming more common as surgical techniques evolve. They are designed to reduce tissue disruption and support faster recovery compared to traditional approaches.
Can spine cage implants be customized?
Yes, custom spine cage manufacturing allows implants to be tailored to a patient’s anatomy. This approach is useful in complex cases where standard designs may not fit properly.
Conclusion
Spine cage manufacturing has evolved into a highly advanced field combining engineering, material science, and medical knowledge. From titanium spine cage designs to PEEK spinal cage innovations, these implants play a key role in modern spinal procedures. Ongoing developments in additive manufacturing and minimally invasive systems continue to shape the future of spinal implant cage technology. Understanding how these devices are made and used helps provide clarity on their role in improving spinal stability and patient outcomes.